By Gareth Vaughan
In late January, as New Zealanders were basking in mid-summer, news of a new coronavirus in the Chinese city of Wuhan seemed a long way off and largely insignificant to most of us. Not so for Lewis Gradon, CEO and managing director of medical device maker Fisher & Paykel Healthcare.
Gradon told interest.co.nz that F&P Healthcare started to see increased demand for its products from China during January, initiated its crisis management procedure at the end of January, and started ramping up manufacturing during February. And F&P Healthcare's staff have barely had time to stop and catch their collective breath since.
Gradon and his team even received early insight into COVID-19 from staff on the ground in Wuhan during January, including sales representative Tina Cui, a former nurse. She visited five hospitals in Wuhan, helping provide F&P Healthcare equipment and train staff on how to use it.
"We had a couple of people in Wuhan during the crisis. I think that's one thing that helped us respond quickly was that we were getting practically first hand knowledge of what was happening in Wuhan at the time. That started in January," Gradon says.
"At the time we were thinking 'gee the whole world's going to look like Wuhan, it's just a matter of time.' One thing we hadn't really accommodated in that thinking was the effectiveness of lockdowns and the way the world's responding. But we certainly saw it first hand."
So what exactly does F&P Healthcare make that's used for treating patients with COVID-19?
"We make a humidifier that goes on these ventilators that you've been hearing about. These ventilators have a tube that goes right down into the lungs and what our system does is it connects the ventilator to that tube. And it makes sure that that gas is at body temperature saturated, so it's at 37º Celsius and 100% relative humidity before it enters the lungs. And we're the world's largest supplier of those humidifiers," Gradon says.
"The second thing we do is a newer therapy. It's called nasal high flow, our system's called Optiflow. And over the course of the pandemic clinical practice has changed. It started off with using the ventilators as first choice. And over time we learned that nasal high flow is a very, very effective way of preventing that ventilator use and getting people better in a much less invasive and much less risky way. And in a way that can preserve ICU beds and ventilators for the patients that really, really need them... Once again we're the world's largest supplier of that nasal high flow therapy."
"We felt quite big responsibilities for our business in that we're the world's largest supplier of one of the two primary treatments for COVID," adds Gradon.
Social distancing and ever increasing work streams
In terms of enacting its crisis management plan in late January, Gradon says as a medical device maker F&P Healthcare can't allow customers to run out of stock.
"So we normally run with a 12 to 14 week pipeline of inventory, so that's the first defence. The second thing we think about is we're a continually growing business so we don't want to be running our plants at 100% of their capacity. So we generally have a manufacturing capacity, as a general rule, a couple of years ahead of what we think our growth will be, that's the second thing we do. Third thing is that, again as a business that's continually growing, we have a manufacturing plan that goes out five years so we almost always have manufacturing on the way. And we make sure that we build our manufacturing buildings in advance so we make sure we've got space."
The unprecedented challenges of COVID-19, however, included setting up a control room that catered for social distancing between staff, and growing out from one workstream.
"Our crisis management plan envisaged one workstream managing the crisis, and that's how it started. And in our crisis management plan we have alternates for all of the roles. Within a couple of weeks we moved to two workstreams. And to cut a long story short, we ended up with a total of 10 different workstreams going all in socially distanced areas," Gradon says.
"One thing that really worked for us, and enabled us to do that, is being in large buildings where we had space to set up very large control rooms where you can get 10 or 20 people in them social distancing."
Keeping supply chains for 1500 parts open
And what about the challenges of manufacturing and making sure a global supply chain for parts and distribution holds together during a global pandemic? Gradon says F&P Healthcare has hired about 700 new manufacturing staff in New Zealand, and about 500 in Mexico, the second country where it has manufacturing facilities, off a base of about 2000 pre-pandemic.
"It has been absolutely massively challenging especially through March, April, May. It kind of started in February. By about a month ago our output of hardware was up about fourfold and that's a steady ramp from about February. It's all very well having manufacturing capacity, but when you're dealing with every single supplier, and for those devices you're talking about around about 1500 components in each piece of hardware. When you're dealing with all these suppliers and you're trying to say 'I want four times, five times, six times the volume I used to have and I want it now,' that's challenging immediately in and of itself," Gradon says.
"And then you add onto that countries going into lockdown, and shutting their borders and shutting down the industries that supply these raw components. It was a pretty common trend around the world. They'd go into lockdown, they'd shutdown, and then it would take a week or two, and it would take government to government intervention sometimes to try and get the medical devices and the raw materials for the medical devices flowing through those borders."
"So it was amazingly busy and that whole supply chain team of ours, getting raw materials in, those guys worked just massive hours. We allocated a whole other group of our research and development people to help them out on that effort. It was pretty much a 24/7 effort," Gradon says.
"And then the other problem we faced was as our supply chain has become depleted, we're airfreighting product around the world from New Zealand and from Mexico. And as everyone probably knows airfreight disappeared at one stage down to 1% of its normal volume. So that was also a daily challenge, just getting the products onto planes."
At times F&P Healthcare sought government assistance to help get its supplies. Gradon says both the Ministry of Foreign Affairs and Trade and New Zealand Trade and Enterprise were "amazingly helpful."
"Every time we needed some help government to government we'd say 'can we have a letter to get this company back in business so we can get the parts for the medical devices you need to treat COVID? Can we have a letter to get our products straight through the border, they're being held up getting in,' things like that. Both government departments were absolutely amazing and everything we needed we generally got it from our government, we got it within 24 hours," Gradon says.
F&P Healthcare's suppliers are located all around the world, he says, with China both a big supplier and a big market, but suppliers are also located across Europe, in the US, and elsewhere in Asia.
In the company's recently released annual report chairman Tony Carter says the COVID-19 pandemic has highlighted how important it is to maintain manufacturing operations in multiple locations.
"Having facilities in both New Zealand and Mexico has been a significant advantage and has given us a higher level of confidence in our business continuity plans. We will continue to progress our work on increasing our manufacturing footprint," Carter says.
Mexican staff get COVID-19
Another challenge for F&P Healthcare is staff at its Tijuana manufacturing facility catching COVID-19.
"COVID is running rampant around communities in Mexico and our facility in the town of Tijuana is no different. Public testing's not available so for our people we funded private testing. And whenever it was available and possible we funded private healthcare for our people," says Gradon.
He estimates more than 50 staff have tested positive, with 33 or 34 now recovered.
"It's an ongoing issue, something we have to manage."
Gradon says a key impact of the COVID-19 pandemic on F&P Healthcare is bringing forward, or speeding up, its growth.
"[We are] installing a lot of hardware in hospitals all around the world. We would have expected that to happen anyway, a bit more slowly. The impact of COVID on our business is we're pulling forward some of what we would've done anyway. I guess it depends how long COVID goes on for. If it goes on for the rest of the year we'd probably think of it as we've just got three or four years ahead. We'll be where we would've been in three or four years time."
Like the rest of us Gradon doesn't know how the pandemic will play out from here.
"We can't predict what happens with COVID around the world, everything's happening kind of for the first time. And we seem to get news every day that might change your view. So we've said we can't predict what COVID does around the world. We can't predict what COVID does for demand on our products. So what we're going to do is we're just going to continue increasing our manufacturing capacity," says Gradon.
"Our current plan has us increasing our manufacturing capacity right up to the end of this year. Obviously we'll assess that regularly and we think that's the socially responsible thing to do whatever COVID does. And if that's not what happens we'll call that a bonus. And we would've made two or three years worth of manufacturing equipment and installed it within 12 months instead of over three years. That's how we're thinking about it."
'We kind of realised we had a tiger by the tail'
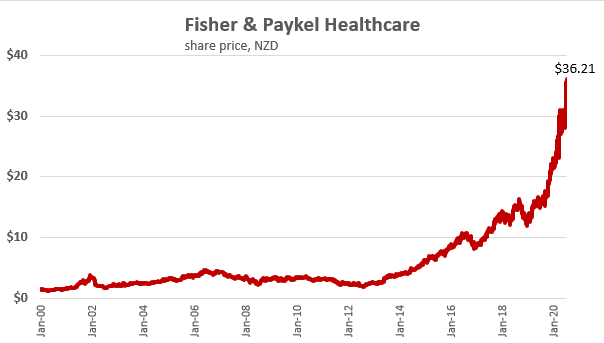
F&P Healthcare was started within Fisher & Paykel Industries, which split into two independent share market listed companies in 2001 with the other being whiteware maker F&P Appliances. F&P Healthcare recently posted a 37% jump in March year profit after tax to $287.3 million, with revenue up 18% to $1.26 billion. The company's market capitalisation recently topped $20 billion and its now the biggest New Zealand share market listed company.
Gradon, who has worked for F&P Healthcare for 37 years, says he didn't think the market capitalisation would top $20 billion a month ago, let alone 30 years ago.
"There was a point in time where we did think 'boy this company's going to go places,' and we kind of realised we had a tiger by the tail. But I don't think we ever imagined until relatively recently that we would be the most valuable company in New Zealand, that's for sure."
The diagrams below come from F&P Healthcare's annual report and an investor presentation.
*This article was first published in our email for paying subscribers. See here for more details and how to subscribe.
16 Comments
Is there a way of honouring such companies as part of Queen's Birthday honours ? Not the CEO alone, the whole company.
Contrarian, err, quiet word to the wise, FPH is a capitalist enterprise owned by the rentier class. You might want to keep such heretical ideas to yourself.
Why ? Capitalists cannot do good or what ? And why keep my ideas to myself ? Freedom of expression and all that, still there. Thanks mate.
No. Capitalism is intrinsically evil. Get with the program !!
I agree with you. NZ culture does not generally respect corporate success. I own a solid chunk of FPH and have enjoyed a very rewarding ride by investing in this fantastic company.
We should certainly be recognising and rewarding such enterprise rather than the easy resort of buying and selling houses to each other for more and more money. FPH is the kind of enterprise of which we need more.
FPH went into crisis mode in Jan and by Feb knew for sure C19 was a malignant monster. Graydon realised by March 'the whole world's going to look like Wuhan, it's just a matter of time'. Why didn't the government? In mid March Ardern is joking with Paddy Gower about his sniffle. She clearly didn't understand the magnitude. FPH is NZ's biggest company. Do the high paid mandarins at MBIE even talk with them ?
Yes but we have added a massive PR paint job over that and Labour is the saviour of everyone and a hell of a lot just swallow it despite knowing differently.
Yes mm my point as well. It goes to show you the gap between the reality at ground zero & the elite-in-charge's arrogance in those early days especially. Australia pushed their emergency button on 27 February. If we'd gone the next week, say the Wednesday after the caucus meeting, we would have 2 weeks ahead of the curve & could have saved taxpayers about 20 billion dollars - that's $20,000,000,000. Go hard & go early was bollocks. They didn't go early that's why they had to go hard.
Mercifully Ardern finally dropped the 'gold standard testing' blather when the penny eventually dropped that it was tosh. But the 'went early' line endures. The princess fan club defend it with the qualification 'based on the available evidence' and while that of itself is debatable (as you outline) this from Graydon shows that if the CoL didn't have the intel that C19 was rampaging and a real existential threat, they damn well ought to have. That someone at high level didn't join the very simple line of dots and give give Louis a call speaks yet again to ministerial incompetence.
This link seems to indicate Australia didn't take significant action until around March 12 .. https://www.mondaq.com/australia/operational-impacts-and-strategy/92962… by that time Australia already had 2 deaths. So don't try and make out that Australia is any better. Not sure when they started closing borders as yet, but NZ started closing borders to China in Early February. And yes we were later in announcing Alert levels on March 21 9 days later. Not sure where you get the 20 billion dollars figure from but if you would rather live in USA then be my guest. And look at where Australia/ Victoria is now, going back in to lockdown. Perhaps you should be more grateful that you live where you do.
I am proud of the fact that my daughter-in-law holds a senior position in the company and did her Masters on nasal oxygen therapy when it was only just beginning to be accepted as legitimate.
I have been a shareholder since 2005.
Well done to her, she's making a real contribution to our little country. You must be very proud. When I started buying FPH back around the same time as you my broker/adviser was negative on them and tried to dissuade me. Very pleased I didn't listen. On a related note, I was interested to see the C19 infection rate for Mexican FPH staff at around 3% (50 of 1500 approx total before current expansion). Official total Mexican infection toll at around that time was 23K from a tot pop of 126m or about .18% if I have my calc right. Which would mean either FPH is a hot bed of infection (unlikely given relative higher socio economic profile of its staff), is a specific non representative cluster or the official community infection rate is significantly higher. Even allowing fora lag in reporting and using say June official infection rate data, the given numbers look hopelessly low if extrapolating the FPH experience.
Given the behaviour of the people in quarantine, the governments lack lustre isolation managment and their abismal track and trace performance (300 odd untraced potentially infected people from the 2 infected funeral atendees fiasco), it is only a matter of time before we have comunity transfer like Melbourne. It is only sensible to assume this and make your personal planning accordingly. How many times can they toss the dice till it comes up double 6 or triple 6.
If I were them I would drastically limit the number of people returning. I note that Australia, with a population of 25 million has just reduced their inflow to 4,000 per week. NZ with a pop'n of 5 million is aiming to increase ours to about this. What is wrong with this picture?
For those people with perment or or temporary residency, they should loose this permanently and be deported if they break quarantine. For born here quarantine breakers, the penalties need to be far harsher and applied without out any lienancy. Applicants wanting to get onto some sort of entry priority list need to sign documentation accepting these conditions.
People need to be locked in their rooms with heavy gaurding for the full duration of Quantine preferably in rooms 3 to 4 stories above ground. For smokers there are nicotine patches. If people do not like these conditions then they do not have to return to NZ. I note that the crew for the Avatar film were never alowed out of their rooms. Why have we now gone weak and sloppy. The cost of these measures are piffling compared to a return to lockdown.
Of course the government will not do this as it is too busy being "kind" to exert any real spine. (the virus does not give a fig about kindness and will immediately exploit any weakness in our defences) When the enevatable occurs, I wonder how kind the public will think they are when they put us back into lock down again or worse still just let the virus propogate. They will thouroughly deserve a sharp reverse in the polls. We all saw how quickly popularity can change when Jacinda came to power. That can work both ways.
For a share that was at around under $2 as recently as July 2012 , a small $150,000. investment would have made you a multi-millionaire today , excluding dividends received .
Easier than speculating in real estate or dealing with Auckland housing tenants that's for sure
Interesting point you make Boaty, about the comparison with housing value gains. I haven't the temperament or interest in being a landlord but realised I needed to have a stake in housing (in addition to homes occupied by us) or miss the gravy train so opted for retirement village shares as a proxy. Over 10 years that has yielded a nearly six times multiple capital return on outlay. By comparison house prices rose by around 60% over the same period. Not apples for apples and tax efficiency needs to be factored but is nevertheless a compelling alternative story.
The thing is company performance and increase in stock prices could never be predicted well. On the other hand, houses had their stable growth and income.

We welcome your comments below. If you are not already registered, please register to comment
Remember we welcome robust, respectful and insightful debate. We don't welcome abusive or defamatory comments and will de-register those repeatedly making such comments. Our current comment policy is here.